At PhoenixBio, we strive to help improve human health through the broad application of our humanized liver chimeric mouse model, the PXB-mouse. Our chimeric mouse model has a highly humanized liver, with human-specific metabolism pathways and gene expression as well as human-like lipid profiles, making this a relevant model for drug discovery and development projects. While we commercially produce PXB-mice using a single human hepatocyte donor lot, we understand that some research may require different hepatocyte donors, such as donors with specific characteristics (HLA typing or disease state) or even donors from different species (NHPs, humans, or others). Therefore, we offer custom transplantation services with our host mouse (cDNA-uPA/SCID background) which allows researchers to select and test engraftment of a hepatocyte donor that meet their specific research needs. First, we will highlight research that used a hepatocyte donor transplanted by our expert team into host animals for a rare genetic disease, Ornithine Transcarbamylase Deficiency (OTCD).
In addition to custom transplantation, there are other ways that the PXB-mouse can be “customized” to enhance your research. For example, the PXB-mouse can be used as a diet-induced disease model, which is an important tool for metabolic liver diseases. Compared to other rodent diet-induced disease models, the PXB-mouse has a human specific metabolism profile which is vastly different from wild type mice. Here, we will also show evidence that our chimeric mouse model can be used to investigate fatty liver disease development and is a relevant model for translational drug discovery and development.
Our PXB-mice have >70% human hepatocyte engraftment and display human-specific metabolism and excretion pathways. While our model recapitulates human outcomes for many therapeutics, some predictions may be improved by suppressing the few residual mouse hepatocytes. To wrap up, we will highlight a recent study that used this approach as another example of how the PXB-mouse can be “customized”.
Custom transplantation to achieve a translational liver disease model
Ornithine transcarbamylase (OTC) is an important enzyme involved in the urea-cycle which is predominately expressed by hepatocytes. OTC deficiency (OTCD) is a metabolic X-linked genetic disease which affects the urea-cycle causing a buildup of toxic ammonia in the blood. While this is the most common urea-cycle disorder there is phenotypic variability in individuals suffering with OTCD1.
Treatment regimens primarily target high ammonia levels with severe cases ultimately needing a liver transplantation. However, new gene therapeutics are under development to target the dysfunctional OTC gene. Testing the efficacy of these new therapeutics requires an animal model. While there are mouse models of hyperammonemia, such as OTCspf-ash, they lack human-specific metabolism pathways and the human OTC gene. Therefore, using a custom transplantation approach in cDNA-uPA/SCID mice to generate humanized OTCD mice offers a novel model.
In a recent study, two OTCD patient donor hepatocytes were transplanted into female homozygous cDNA-uPA/SCID mice to generate a humanized OTCD model2. Human albumin (h-Alb) levels were low in mice engrafted with patient hepatocytes so to improve the engraftment serial transplantation methods were used which significantly increased the blood h-Alb levels. The repopulation of hepatocytes was improved in serially transplanted OTCD mice (Figure 1), and patient hepatocytes expanded to occupy the host mouse liver tissue. Additionally, these mice exhibited significant increases in blood ammonium levels, specifically in later generation OTCD mice, and decreased OTC enzyme activity (Figure 2). 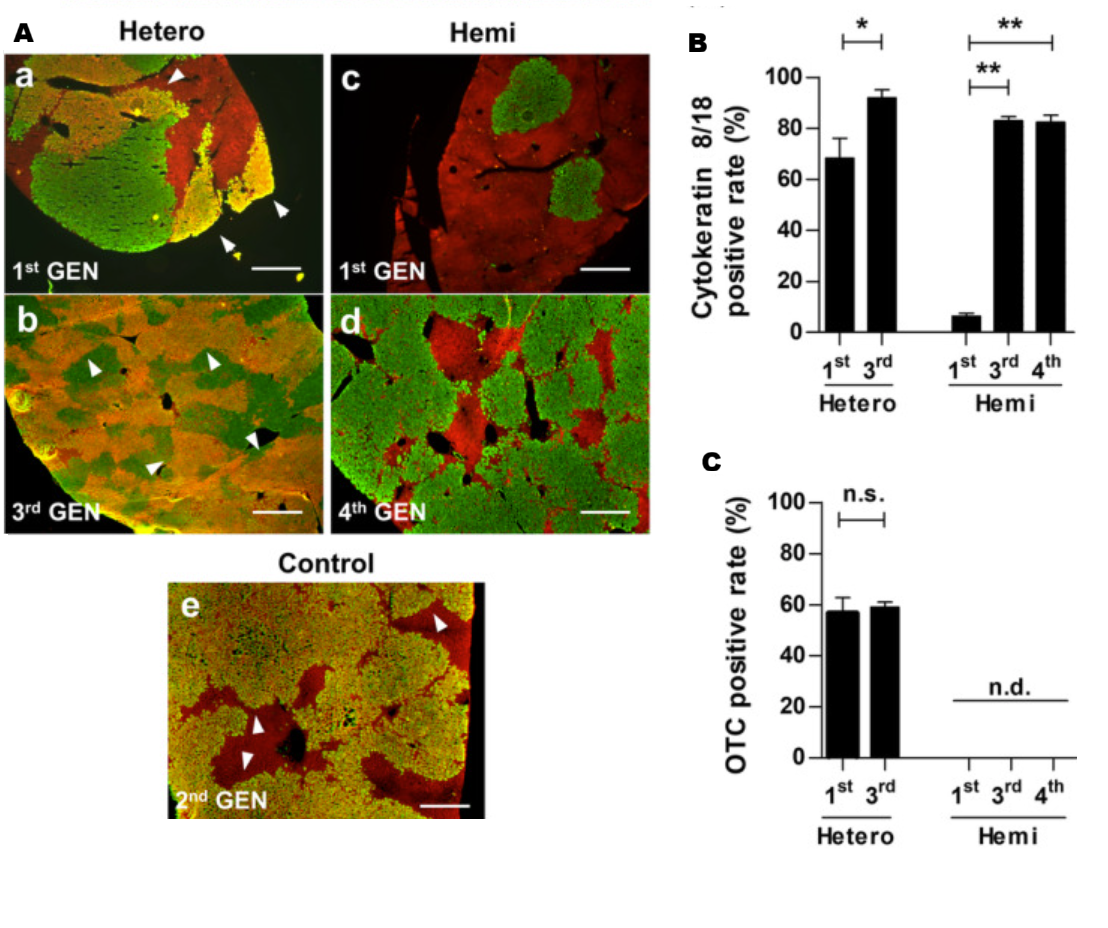
Figure 1: Two patient OTCD hepatocyte donors were used to generate OTCD humanized mice, one with heterozygous OTC mutation (Hetero) and one with a hemizygous OTC mutation (Hemi). A. Staining for human cytokeratin 8/18 (green) and OTC (red) a-b hetero OTCD 1st and 3rd generation, c-d hemi OTCD 1st and 4th generation, and e control PXB-mice. B. Percent of area positive for hCK8/18 C. Percent of area that is double-positive for OTC and hCK8/18. n.s = not significant, n.d. = not detected. (Adapted from Sugahara et al., 2020)
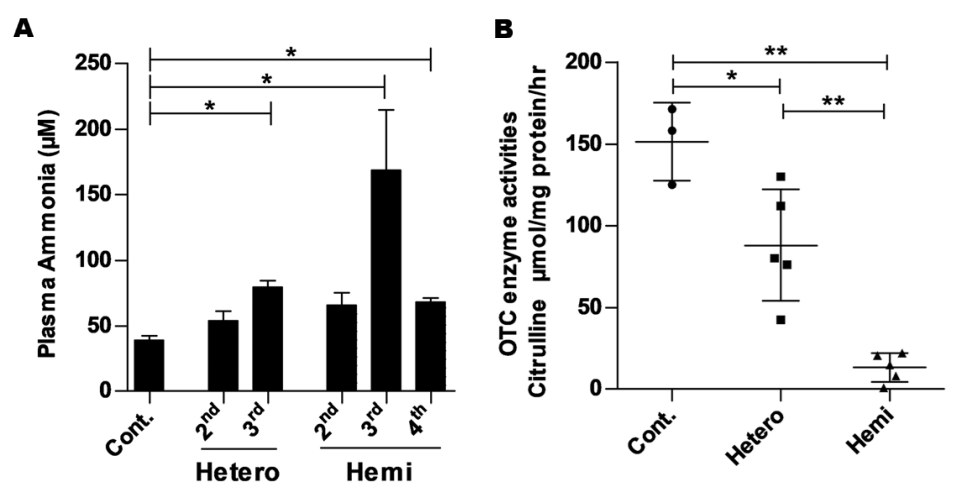
Figure 2: OTCD mice had buildup of ammonia with decreased OTC enzyme activity. A. Plasma ammonia levels in OTCD mice (Hetero and Hemi) and control PXB-mice B. OTC enzyme activity was assessed in isolated hepatocytes from hetero-OTCD, hemi-OTCD, and control PXB-mice. (Adapted from Sugahara et al., 2020)
This research demonstrates the versatility of our cDNA-uPA/SCID mouse, not only allowing us to transplant healthy human hepatocytes (PXB-mice), but also diseased hepatocytes with success. Our custom transplantation service offers the ability to tailor your ideal chimeric mouse model.
Diet induced fatty liver disease in the PXB-mouse model
Metabolic dysfunction-associated steatotic liver disease (MASLD) and metabolic dysfunction-associated steatohepatitis (MASH) cases have steadily increased over the years and are projected to continue to rise3. MASLD and MASH are associated with an increased risk of comorbidities and can lead to liver cirrhosis and hepatocellular carcinoma (HCC).
Due to the increased incidence of MASLD it is important to understand the underlying pathogenesis and to develop novel therapeutics to prevent the progression and ultimately reverse the associated damage of MASLD/MASH. Pre-clinical mouse models often use diet-induced MASLD however, due to the metabolic differences between mouse and human hepatocytes these models might not fully recapitulate the disease progression and/or predict the clinical relevance of therapeutics. Therefore, inducing fatty liver disease in the PXB-mouse is a promising model to test clinical drugs in development.
Two diets that can be used to induce fatty liver include the Gubra Amylin NASH (GAN) diet, which is a high-fat, high-fructose, and high-cholesterol diet, and the choline-deficient, amino acid-defined, high fat diet (CDAHFD). PXB-mice fed the GAN diet have significant increases in the liver/kidney ratio which is used to estimate fatty liver incidence (Figure 3)4.
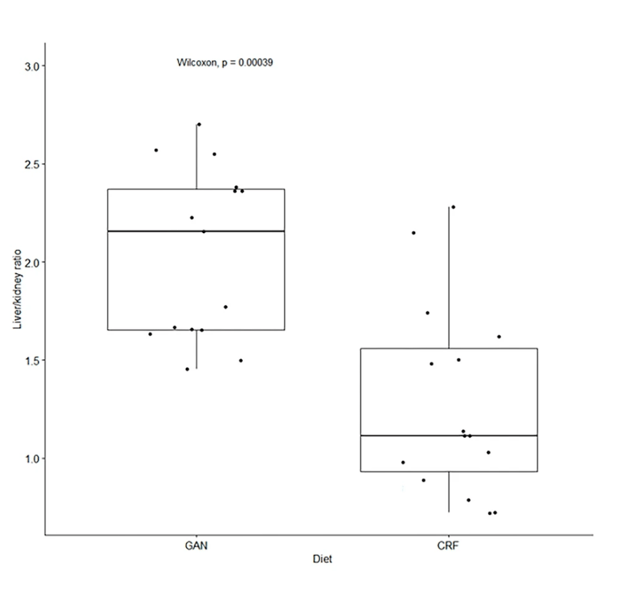
Figure 3: Fatty liver was estimated by ultrasound, using the liver to kidney ratio. GAN-fed PXB-mice had significantly higher liver/kidney ratios than control diet mice. (Adapted from Ichikawa et al., 2024)
CDAHFD-fed PXB-mice have large lipid droplet accumulation in the liver and there are Mallory-Denk body-like aggregates observed in ballooning hepatocytes (Figure 4A-B)5. Additionally, fibrosis was increased in CDAHFD-fed mice compared to control animals (Figure 4C-D) and α-smooth muscle actin-positive cells was increased (Figure 4E), suggesting hepatic stellate cell activation was achieved in this model. Importantly, there was marked improvement in steatosis and ballooning in PXB-mice treated with Elafibranor in a prophylactic context (Figure 5).
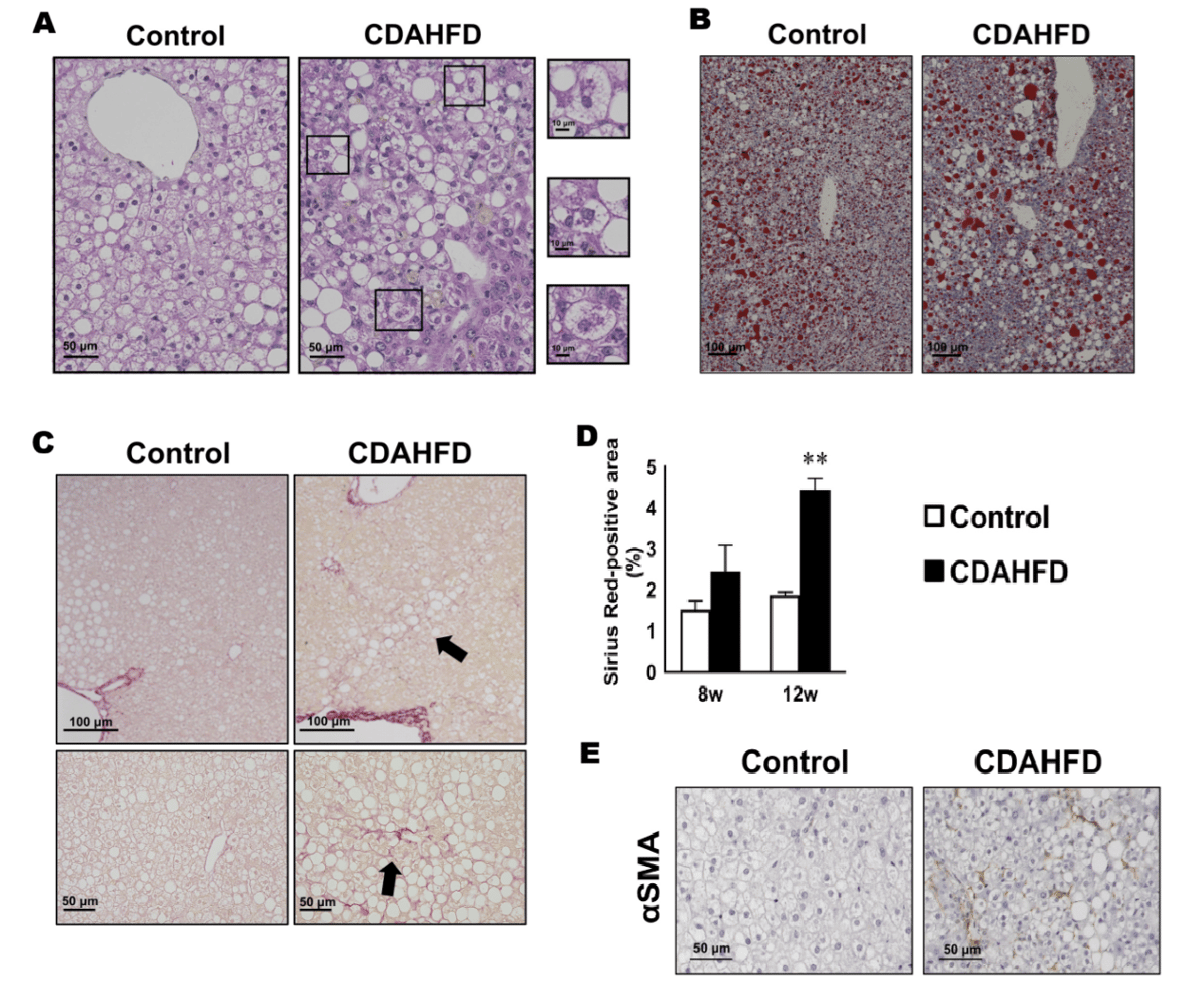
Figure 4: Large lipid droplets were observed in CDAHFD-fed PXB-mice A. Liver H&E staining of PXB-mice fed either control or CDAHFD for 12-weeks. Magnified areas show ballooning hepatocytes with aggregates (Mallory-Denk body like aggregates). B. Oil Red O staining. C. Fibrosis was determined by Sirius Red staining D. Ratio of Sirius red-positive areas E. α-smooth muscle actin (αSMA). (Adapted from Kisoh et al., 2024)
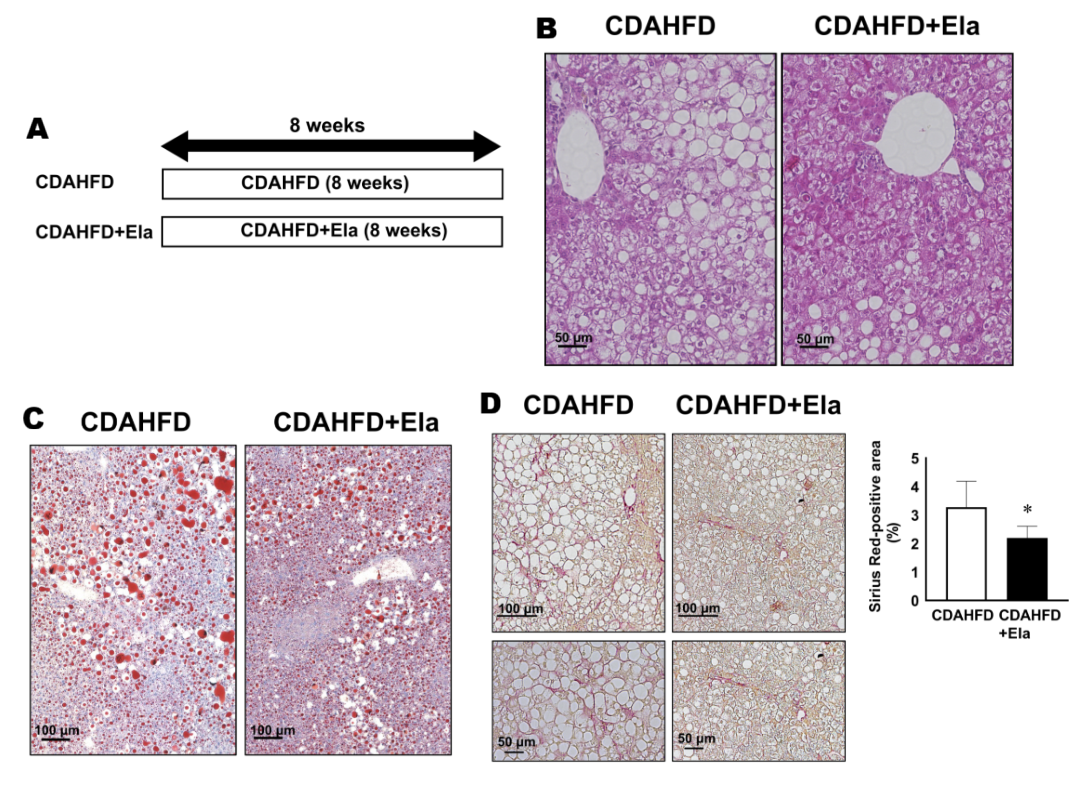
Figure 5: Elafibranor treatment reduces progression of steatosis and hepatic ballooning A. Study design B. Liver H&E staining C. Oil Red O staining and D. Sirius red staining of treated or untreated CDAHFD-fed mice (Adapted from Kisoh et al., 2024)
Taken together, PXB-mice are suitable for diet-induced MASLD/MASH, demonstrating the broad adaptability of this humanized liver model.
Using species-specific siRNA to alter gene expression of PXB-mice
While the PXB-mouse is highly humanized there are residual mouse hepatocytes in this model. Due to species differences in metabolism, specifically cytochrome P450 (CYP)-mediated metabolism, these diseased mouse hepatocytes may affect the pharmacokinetic (PK) profile of PXB-mice to some degree. In a recent study, Dr. Yamazaki and colleagues aimed to further reduce the impact of residual mouse hepatocytes by using targeted silencing of murine CYP oxidoreductase (POR) gene (Figure 6)6. Figure 6: Graphical representation of how targeted siRNA can alter the metabolism of PXB-mice. (Adapted from Yamazaki et al., 2025)
Figure 6: Graphical representation of how targeted siRNA can alter the metabolism of PXB-mice. (Adapted from Yamazaki et al., 2025)
They observed a significant reduction in mouse specific POR siRNA-LNP treated PXB-mice with no effect on human POR gene expression (Figure 7). To test the effect on metabolism, PXB-mice were then treated with Warfarin. The Warfarin metabolite profile of PXB-mice (saline) is more similar to human liver microsomes than mouse liver microsomes (Figure 8). However, reducing mouse POR gene expression further accentuates the human metabolism of PXB-mice (Figure 8).
 Figure 7: siRNA targeting mouse POR gene significantly reduces the mouse gene expression without affecting human POR. (Yamazaki et al., 2025)
Figure 7: siRNA targeting mouse POR gene significantly reduces the mouse gene expression without affecting human POR. (Yamazaki et al., 2025)
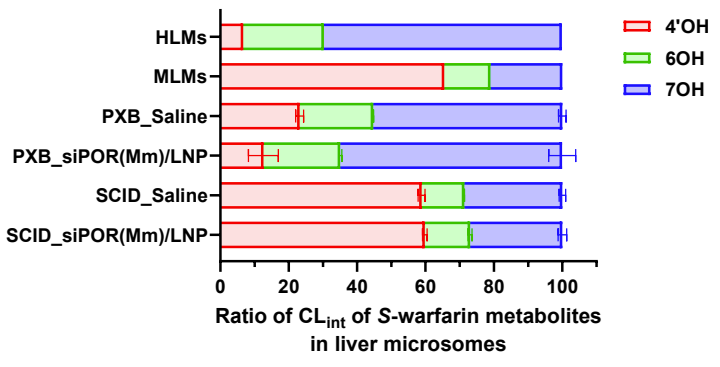
Figure 8: Warfarin metabolite ratios of human liver microsomes (HLMs), mouse liver microsomes (MLMs), untreated PXB-mice (PXB_Saline), targeted siRNA treated PXB-mice (PXB_siPOR(Mm)/LNP), untreated SCID (SCID_Saline), and targeted siRNA treated SCID (SCID_siPOR(Mm)/LNP). (Yamazaki et al., 2025)
Refining the PXB-mouse by using targeted gene silencing of the residual mouse hepatocytes could be advantageous for some PK studies that are impacted by mouse CYP activity.
Conclusion
From custom transplantation to targeted refinement of PXB-mice, chimeric mice with a humanized liver are a valuable translational research model advancing drug discovery and development. Our expert staff have over 15 years of expertise in generating chimeric mice and are eager to help you plan your customized study, contact us today.
Contact us to find out more about how cryopreserved PXB-cells can support your research!
References
-
Seker Yilmaz B, et al. Liver transplantation in ornithine transcarbamylase deficiency: A retrospective multicentre cohort study. Mol Genet Metab Rep. (2023) 37:101020. doi:10.1016/j.ymgmr.2023.101020
-
Sugahara G, et al., Humanized liver mouse model with transplanted human hepatocytes from patients with ornithine transcarbamylase deficiency. J Inherit Metab Dis. (2020) 44(3):618-628 doi: 10.1002/jimd.12347
-
Le P, et al., Estimated burden of metabolic dysfunction-associated steatotic liver disease in US adults, 2020 to 2050. JAMA Netw Open. (2025) 8(1):e2454707. Doi: 10.1001/jamanetworkopen.2024.54707
-
Ichikawa A, et al., Multi-omics analysis of a fatty liver model using human hepatocyte chimeric mice. Scientific Reports. (2024) 14:3362 doi: 10.1038/s41598-024-53890-8
-
Kisoh K et al., Estimating drug efficacy with a diet-induced NASH model in chimeric mice with humanized livers. Biomedicines. (2021) 9(11):1647 doi: 10.3390/biomedicines9111647
-
Yamazaki K, et al., Species-specific gene expression manipulation in humanized livers of chimeric mice via siRNA-encapsulated lipid nanoparticle treatment. Mol Ther Methods Clin Dev. (2025) 33(2):101466. doi: 10.1016/j.omtm.2025.101466

