New approach methodologies (NAMs) are innovative, non-animal scientific methods used for testing chemical safety, drug development and biological research. In 2025, the FDA announced a new initiative to phase out animal testing for certain types of drug development1. This has led to renewed interest in NAMs and their role in pre-clinical drug development. NAMs include advanced in vitro models such as co-culture models, organ on a chip, 3D bioprinting and tissue models and Microphysiological systems (MPS)
PXB-cells are high quality, primary human hepatocytes that are derived from a proprietary in vivo bioreactor. They are highly plateable and have excellent characteristics for long term cell culture, routinely used for experiments in the range of 21-28 days. Moreover, they are validated for transporter and CYP enzyme activity2,3. Here we will discuss how PXB-cells can be applied to develop NAMs and provide high quality pre-clinical data.
Investigating the impact of the liver microenvironment on hepatocyte function
Liver architecture is key to its function. Modeling hepatic function in vitro requires consideration of features such as zonation, interaction with other cell types, blood vessels, and bile ducts4. Proximity to blood vessels impacts the delivery of oxygen to hepatocytes leading to heterogeneity of expression of many genes, including metabolic enzymes. Moreover, mechanical forces from blood flow may also impact liver function. Thus, it is important to consider these parameters when developing in vitro liver models. A recent study examined the impact of oxygen concentration on hepatocyte (PXB-cells) function in monoculture and in co-culture with liver sinusoidal endothelial cells (LSECs)5. In addition, a dynamic co-culture system which incorporated media flow was also generated to mimic interstitial shear stress experienced by hepatocytes in vivo (Figure 1). Oxygen concentrations of 5% or 10% were used to mimic the pericentral and periportal regions of the liver, respectively (Figure 1)5.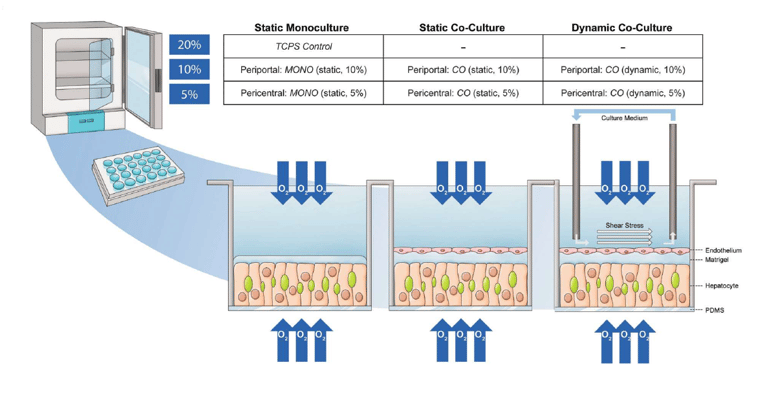
Figure 1: Experimental setup to investigate the contribution of microenvironmental parameters (O2 concentration, shear stress) on hepatocyte (PXB-cells) monocultures, or co-cultures with liver sinusoidal endothelial cells (LSECs). (Adapted from Scheidecker et al., 2023)
The results showed that the activity of drug-metabolizing enzymes was increased under static culture conditions with physiological oxygen levels of 5-10% (Figure 2)5.
 Figure 2: Metabolic stability assay of substrates with known primary drug metabolism shows that CYP450 activity is overall better maintained in static co-culture. (Adapted from Scheidecker et al., 2023)
Figure 2: Metabolic stability assay of substrates with known primary drug metabolism shows that CYP450 activity is overall better maintained in static co-culture. (Adapted from Scheidecker et al., 2023)
Interestingly, the dynamic co-culture system created an environment that was more conducive to liver tissue regeneration, indicated by changes in the metabolite profiles (Figure 3A)5 and by the prevalence of inflammatory and cell motility pathways when metabolomic and transcriptomic data was integrated using ingenuity pathway analysis (IPA) (Figure 3B)5.
 Figure 2: A) Global metabolomic analysis comparing co- and mono-cultures with varying oxygen concentrations under static or dynamic conditions reveals distinct metabolite accumulations. B) IPA analysis of the dynamic vs static monocultures under different oxygen conditions reveals that inflammatory and cell motility pathways are more active in the dynamic cultures, which is indicative of hepatocytes in a regenerative state. (Adapted from Scheidecker et al., 2023)
Figure 2: A) Global metabolomic analysis comparing co- and mono-cultures with varying oxygen concentrations under static or dynamic conditions reveals distinct metabolite accumulations. B) IPA analysis of the dynamic vs static monocultures under different oxygen conditions reveals that inflammatory and cell motility pathways are more active in the dynamic cultures, which is indicative of hepatocytes in a regenerative state. (Adapted from Scheidecker et al., 2023)
These findings support the use of PXB-cells in co-culture models and highlight that in vitro systems are not one size fits all; even when working within the same tissue it is critical to validate the model for the application under investigation.
Vascularized Liver Models for DILI
Drug induced liver injury (DILI) is a key reason why new therapeutics fail to reach market. Sinusoidal obstruction syndrome (SOS), previously known as veno-occlusive disease is a type of liver injury arising from damage to the blood vessels of the liver6. It is a potential complication of liver transplant and hematopoietic stem cell transplant, potentially as a result of cytotoxic conditioning therapies that damage LSECs. Developing in vitro vascularized liver models are important to identify new therapeutics that might cause this type of liver injury. To address this, Naito and colleagues used a technique called CAViTs to create an artificial liver tissue that is a tri-culture hepatic vascular model7. It includes PXB-cells (hepatocytes), hepatic stellate cells and LSECs. The resulting three-dimensional tissue model is referred to as a VHT (Vascularized Hepatic Tissue) (Figure 4A)7. Initial characterization using immunofluorescent staining and gene expression analysis found that the functionality of the three cell types was maintained or improved in the VHT (Figure 4B)7. Figure 4: (A) Image of the fabrication of a viscous body and the process of generating vascularized hepatic tissue (VHT). (B) Confocal laser scanning microscope (CLSM) images of CD31 (purple) and albumin (green) for VHT on day 6. To the right are enlarged views of the images. Scale bars are 500 μm (left) and 100 μm (right). (Adapted from Naito et al., 2022)
Figure 4: (A) Image of the fabrication of a viscous body and the process of generating vascularized hepatic tissue (VHT). (B) Confocal laser scanning microscope (CLSM) images of CD31 (purple) and albumin (green) for VHT on day 6. To the right are enlarged views of the images. Scale bars are 500 μm (left) and 100 μm (right). (Adapted from Naito et al., 2022)
To validate VHTs for DILI detection, a number of compounds known to induce liver injury were tested. Using margin-of safety (MOS) values obtained by dividing IC50 by Cmax, VHTs registered the toxic effects of troglitazone, acetaminophen and cyclophosphamide better than 2D culture. Non-DILI-associated compounds (minoxidil, flumazenil, phentolamine, pyridostigmine) were found to be non-toxic in VHT assay. Overall, VHTs performed significantly better than 2D cultures of just PHHs. The results were more comparable to spheroid cultures, but some differences were observed7. For example, VHTs failed to predict diclofenac toxicity although it was detected in spheroids. However, cyclophosphamide, which does not register as toxic in spheroids, did lead to toxicity in VHTs. This compound is reported to be associated with clinical cases of SOS — perhaps indicating that its mechanism of toxicity is vasculature dependent7,8.
To further validate VHTs as good models to predict vascular injury, pyrrolizidine alkaloid (PA) therapeutics were studied. This class of compounds has previously been shown to induce SOS. Monocrotaline and retrorsine were chosen as representative compounds and compared with nefazodone, an unrelated compound that is not associated with SOS. Clear dose dependent shortening of vessels was observed following exposure to monocrotaline and retrorsine, while high doses of nefazodone was required to show any impact on vascular morphology (Figure 5)7. Overall, this is a useful vascularized model that can provide additional insights into potential vascular toxicity, that may be missed in spheroids or 2D cultures, further demonstrating the versatility of PXB-cells.
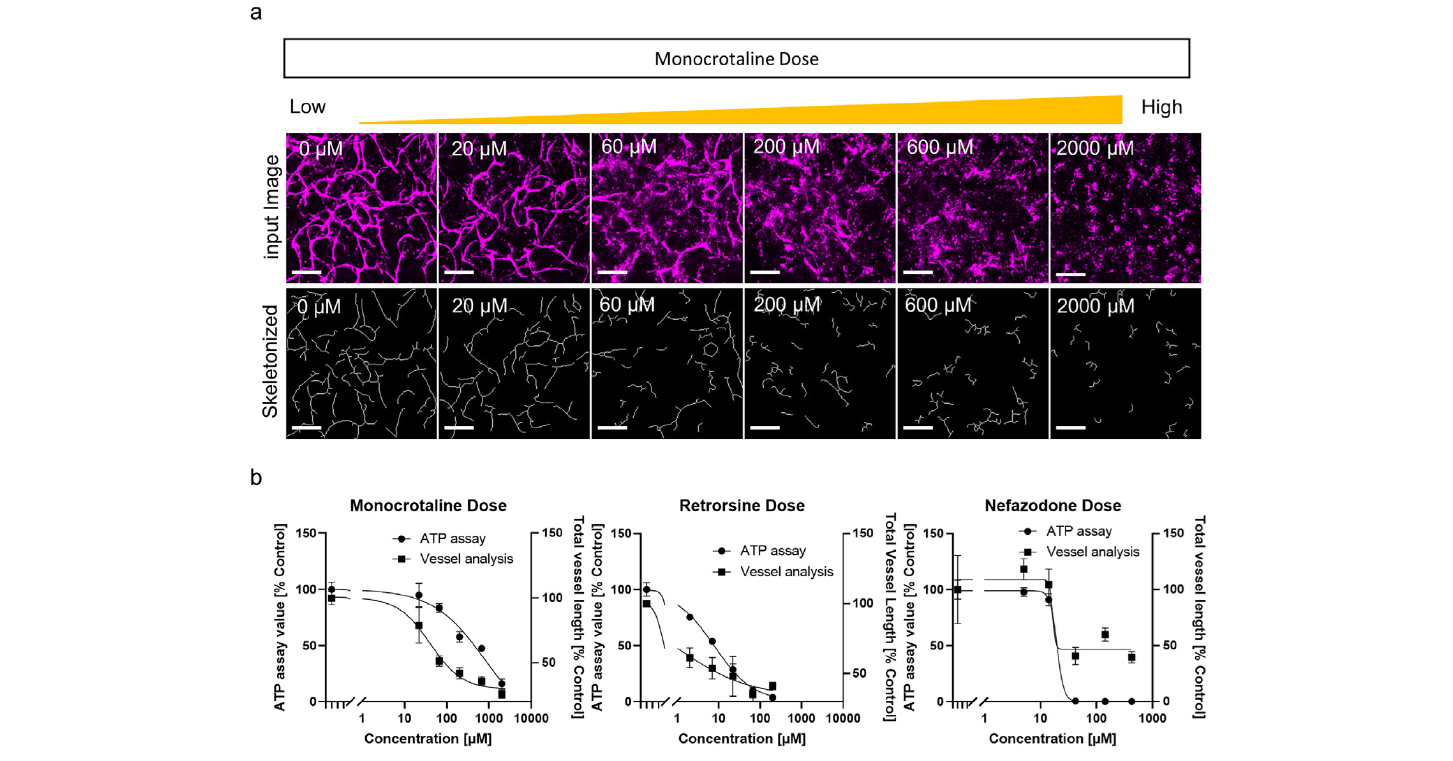
Figure 5: Vessel analysis of compound treated VHT. (a) Images of vessels treated with different monocrotaline doses. Original images (top), and skeletonized images (bottom). Scale bars are 200 μm. Images show monocrotaline dose–dependent damage to vessels. (b) Representative dose response curves for monocrotaline, retrorsine, and nefazodone in the ATP assay or vessel analysis. (Adapted from Naito et al., 2022)
Modeling Gut-liver Interactions with Microphysiological Systems (MPS)
There is great interest in developing multiorgan microphysiological systems (MPS) with the goal of improving clinical pharmacokinetic (PK) and pharmacodynamic (PD) prediction for novel therapeutics9,10. These systems contain cells from different organ systems to better mimic the complex systems found in vivo. Many therapeutics are absorbed via the gut, thus understanding the interplay between the intestines and the liver is critical for fully evaluating the absorption and metabolism of new compounds. With this in mind, a new MPS was recently developed to model the gut-liver axis using a pneumatic-pressure driven system11. It consists of a co-culture of hiPSC-derived intestinal cells and fresh PXB-cells with medium circulation in an open system to facilitate maintenance of the cells and collection of samples (Figure 6)11. Co-culture with the intestinal-like cells led to significant upregulation of albumin production, cytochrome activity and liver-specific gene expression in the human hepatocytes (Figure 7B, C)11. Improved formation of bile canaliculi was also observed in the co-cultured PXB-cells (Figure 7A)11. Together, this suggests that this system, and other MPS, provides a good basis for investigation of hepatic metabolism.
 Figure 6: Pneumatic pressure-driven two-organ MPS used for co-culture of hiPS-intestinal cells and fresh PXB-cells. (a) Photograph of the MPS. (b) Design of the PDMS microfluidic plate containing eight throughput culture units. Each culture unit consists of the hiPS-intestinal and PXB-cell culture chambers. (c) Schematic of the circulation flow direction during cell co-culture. (Adapted from Shinohara et al., 2021)
Figure 6: Pneumatic pressure-driven two-organ MPS used for co-culture of hiPS-intestinal cells and fresh PXB-cells. (a) Photograph of the MPS. (b) Design of the PDMS microfluidic plate containing eight throughput culture units. Each culture unit consists of the hiPS-intestinal and PXB-cell culture chambers. (c) Schematic of the circulation flow direction during cell co-culture. (Adapted from Shinohara et al., 2021)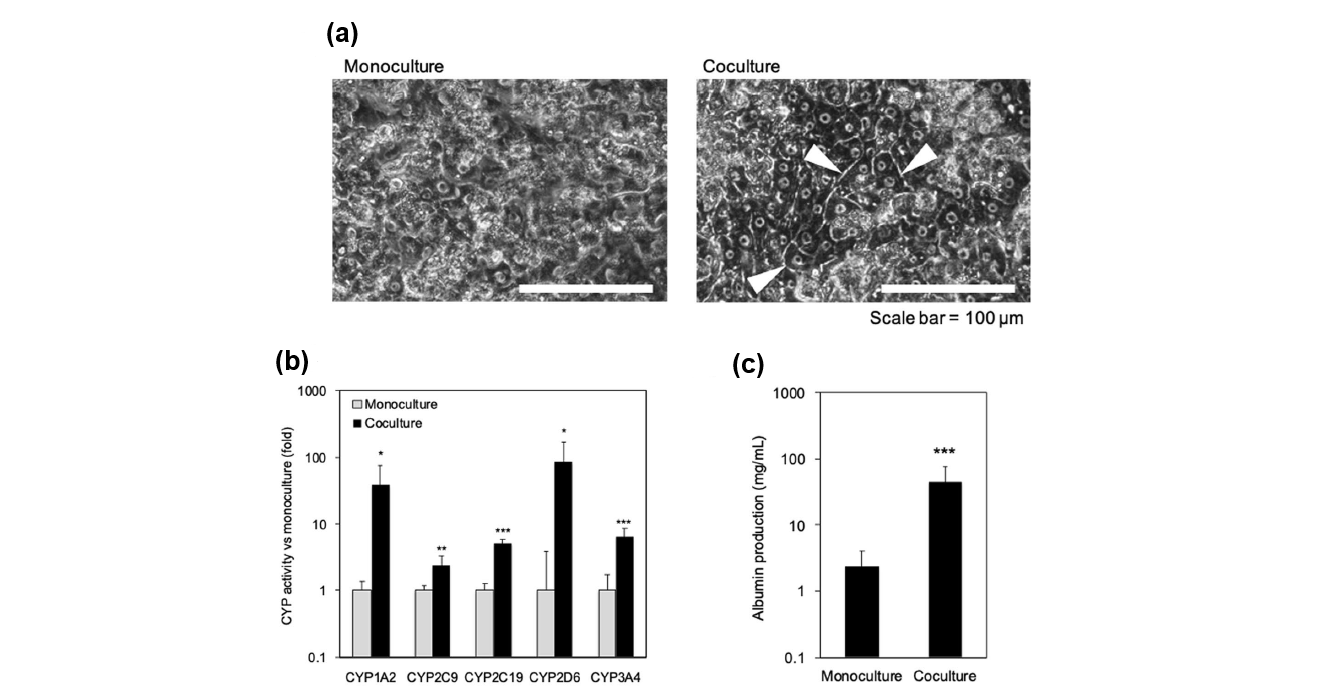
Figure 7: Evaluation on co-culture effects in PXB-cells. (A) Morphology of PXB-cells in monoculture and
Co-culture. The white arrows point to bile canaliculi structures. (B) Enhancement in liver function in PXB-cells
by co-culture with iPS-intestinal cells. The CYP activities using each specific substrate in monoculture
and co-culture of PXB-cells were measured for 48 h. Each bar represents the mean ± SD (n = 8) from two independent experiments normalized to the value obtained from the monoculture. (C) Albumin concentration in culture medium. Each bar represents the mean ± SD (n = 11) from two independent experiments. (Adapted from Shinohara et al., 2021)
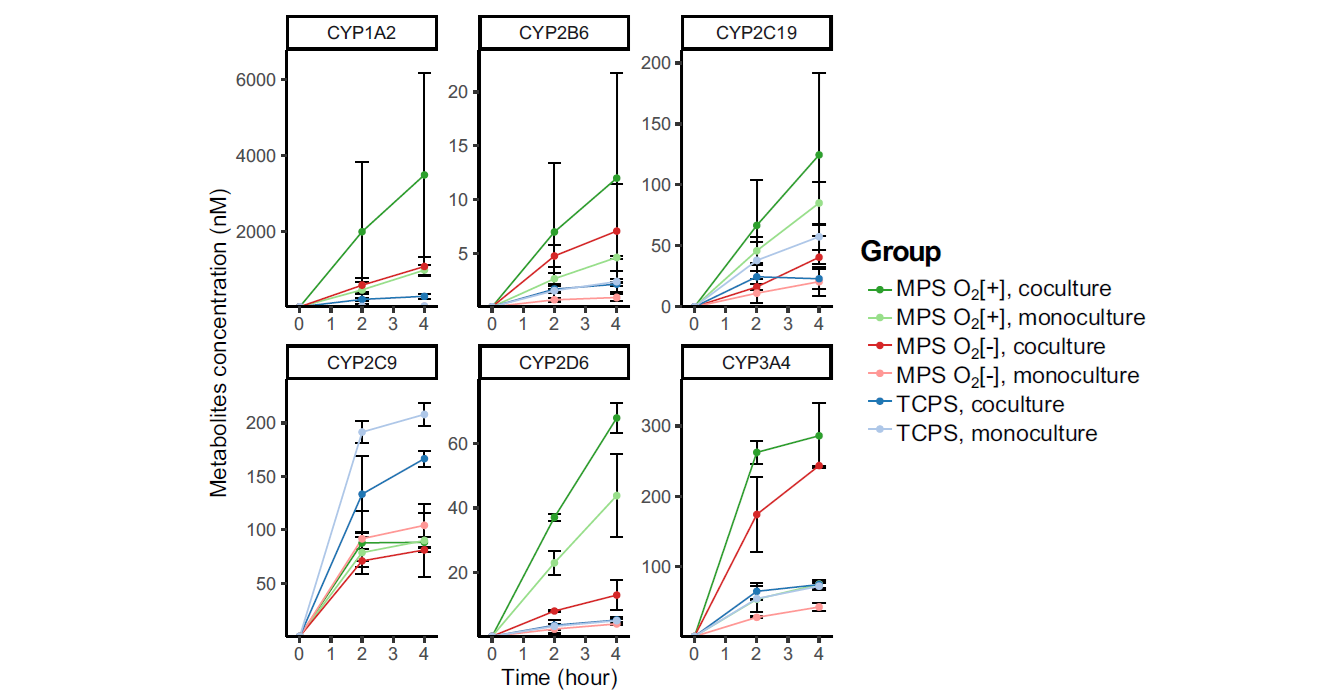
In 2024, Kurniawan and colleagues used this model to investigate new mechanisms of cross-talk between the intestinal cells and hepatocytes12. They examined differentially expressed genes (DEGs) in monocultured or co-cultured cells that were either under perfusion conditions (O2-) or under conditions of perfusion plus direct oxygenation using an oxygen permeable membrane (PMP). They found that CYP enzyme activity was enhanced in the hepatocyte-intestinal cell co-cultures, with a further increase in activity observed in the directly oxygenated group (Figure 8)12.
Figure 8: CYP enzyme activity of PXB-cells measured at the endpoint of 3-day co-culture period. Values show concentration of specific CYP metabolites in the culture supernatant at certain time point (n = 3). MPS= Microphysiological System _+/- direct oxygenation using oxygen permeable membrane as well bottom. TCPS = tissue culture polystyrene plate (conventional cell culture control). Adapted from Kurniawan et al., 2024.
To probe the mechanisms governing these differences, RNAseq experiments were carried out to investigate gene expression changes in the PXB-cells cultured in the various conditions. In the directly oxygenated/MPS group, the most significantly upregulated gene expression was found to be the epoxygenase P450 pathway, which comes under the Arachidonic Acid (ARA) metabolic process. Thus, ARA was tested to see if it could enhance drug metabolism in mono-cultured hepatocytes. Although treatment of PXB-cells with ARA did not significantly augment expression of CYP2C9 or CYP1A2, some minimal increases for other CYPs were observed, suggesting that ARA contributes to the enhancement of hepatocyte metabolism seen during co-culture with intestinal cells, but is not the sole driver of this process12. Overall, MPS models using PXB-cells are useful for studying inter-organ interactions and could also be applied to evaluate the metabolism or liver-directed effects of novel therapeutics.
Conclusion
NAMs promise to reduce or replace animal use in research for many applications that are key to drug development including PK, Toxicology and PD studies. They have the potential to provide data faster and in a more cost-effective manner than traditional in vivo models. Moreover, they could lead to improved prediction of human responses and more human-relevant data at an earlier stage of drug development.
PXB-cells are amenable to inclusion in various types of NAMs and provide a consistent source of primary human hepatocytes that can help with reproducibility. As focus on NAMs increases and more work is done to improve standardization across these emerging models, PXB-cells have high potential to provide a reliable source of well characterized primary human hepatocytes for a variety of applications.
References
- https://www.fda.gov/news-events/press-announcements/fda-announces-plan-phase-out-animal-testing-requirement-monoclonal-antibodies-and-other-drugs
- Sugahara G, Ishida Y, Lee JJ, et al. Long-term cell fate and functional maintenance of human hepatocyte through stepwise culture configuration. FASEB J. 2023;37(2):e22750. doi:10.1096/fj.202201292RR
- Yamasaki C, Ishida Y, Yanagi A, Yoshizane Y, Kojima Y, Ogawa Y, Kageyama Y, Iwasaki Y, Ishida S, Chayama K, Tateno C. Culture density contributes to hepatic functions of fresh human hepatocytes isolated from chimeric mice with humanized livers: Novel, long-term, functional two-dimensional in vitro tool for developing new drugs. PLoS One. 2020 Sep 11;15(9):e0237809. doi: 10.1371/journal.pone.0237809
- Paris J, Henderson NC. Liver zonation, revisited. Hepatology. 2022 Oct;76(4):1219-1230. doi: 10.1002/hep.32408. Epub 2022 Mar 6. PMID: 35175659; PMCID: PMC9790419.
- Scheidecker B, Poulain S, Sugimoto M, Arakawa H, Kim SH, Kawanishi T, Kato Y, Danoy M, Nishikawa M, Sakai Y. Mechanobiological stimulation in organ-on-a-chip systems reduces hepatic drug metabolic capacity in favor of regenerative specialization. Biotechnol Bioeng. 2024 Apr;121(4):1435-1452. doi: 10.1002/bit.28653. Epub 2024 Jan 7. PMID: 38184801.
- Corbacioglu S, Jabbour EJ, Mohty M. Risk Factors for Development of and Progression of Hepatic Veno-Occlusive Disease/Sinusoidal Obstruction Syndrome. Biol Blood Marrow Transplant. 2019 Jul;25(7):1271-1280. doi: 10.1016/j.bbmt.2019.02.018. Epub 2019 Feb 22. PMID: 30797942.
- Naito Y, Yoshinouchi Y, Sorayama Y, Kohara H, Kitano S, Irie S, Matsusaki M. Constructing vascularized hepatic tissue by cell-assembled viscous tissue sedimentation method and its application for vascular toxicity assessment. Acta Biomater. 2022 Mar 1;140:275-288. doi: 10.1016/j.actbio.2021.11.027. Epub 2021 Nov 24. PMID: 34826641.
- Rezvani AR, McCune JS, Storer BE, Batchelder A, Kida A, Deeg HJ, McDonald GB. Cyclophosphamide followed by intravenous targeted busulfan for allogeneic hematopoietic cell transplantation: pharmacokinetics and clinical outcomes. Biol Blood Marrow Transplant. 2013 Jul;19(7):1033-9. doi: 10.1016/j.bbmt.2013.04.005. Epub 2013 Apr 10. PMID: 23583825; PMCID: PMC3898851.
- Chang SY, Weber EJ, Ness KV, Eaton DL, Kelly EJ. Liver and Kidney on Chips: Microphysiological Models to Understand Transporter Function. Clin Pharmacol Ther. 2016 Nov;100(5):464-478. doi: 10.1002/cpt.436. Epub 2016 Aug 27. PMID: 27448090; PMCID: PMC5056140.
- Van Ness KP, Cesar F, Yeung CK, Himmelfarb J, Kelly EJ. Microphysiological systems in absorption, distribution, metabolism, and elimination sciences. Clin Transl Sci. 2022 Jan;15(1):9-42. doi: 10.1111/cts.13132. Epub 2021 Aug 26. PMID: 34378335; PMCID: PMC8742652.
- Shinohara M, Arakawa H, Oda Y, et al. Coculture with hiPS-derived intestinal cells enhanced human hepatocyte functions in a pneumatic-pressure-driven two-organ microphysiological system. Sci Rep. 2021;11(1):5437. Published 2021 Mar 8. doi:10.1038/s41598-021-84861-y
- Kurniawan DA, Leo S, Inamatsu M, et al. Gut-liver microphysiological systems revealed potential crosstalk mechanism modulating drug metabolism. PNAS Nexus. 2024;3(2):pgae070. Published 2024 Feb 9. doi:10.1093/pnasnexus/pgae070